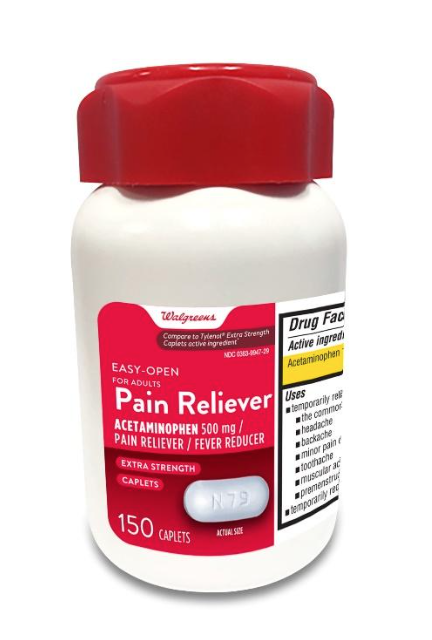
Walgreens Pain Reliever Acetaminophen, 150 count bottles
Description: This recall involves the Walgreens brand acetaminophen product. The red and white label states Walgreens, Easy Open for Adults, Pain Reliever, Acetaminophen, 500 mg, Fever Reducer, Extra Strength, 150 caplets. The bottle has a red continuous thread gear closure. UPC number 311917218090 and Lot numbers P2100627, P2100671, P2100672, P2100689 P2100747, P2100859 (each with expiration date Nov-2022) and P2200050 (with expiration date Jan-2023) are included in this recall. The UPC number, lot numbers and expiration date are printed near the drug facts panel on the label on the back of the bottle. [Learn More]
About 137,300 units were affected by this recall.
Aurohealth Recalls Walgreens Brand Acetaminophen Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning
Recalled Walgreens Pain Reliever Acetaminophen 500 mg, 150 Count Bottle. The UPC code, lot number and expiration date can be found on the label on the back of the bottle
Walgreens Pain Reliever Acetaminophen, 150 count bottles Recall Information
| Country of Origin | India |
| Recall Date | 06/16/2022 |
| Recall ID | 9327 |
| Recall Number | 22161 |
| Hazard | The recalled over-the-counter products contain the regulated substance acetaminophen which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children. |
| Injuries | None reported |
| Remedy | Consumers should immediately store the recalled products in a safe location out of reach and sight of children. Contact Aurohealth for information on how to return the product at your nearest Walgreens store to receive a full refund. |
| Remedy Option | Refund |
| Contact | Aurohealth toll-free at 888-504-2014 from 8 a.m. to 5 p.m. CT, Monday through Friday, online at www.walgreens.com/topic/information/recall.jsp, www.walgreens.com and click on "product recalls" at the bottom of the page, or www.aurohealth.com and click on "RECALL" at the top of the page for more information. |
| Last Updated | 06/16/2022 |
| Similar To |
