Children’s Pain & Fever Concentrated Drops
Description: The recall involves Rugby Children's Pain & Fever Concentrated Drops (Acetaminophen Drops) in a 1/2 fl. oz. (15 ml) bottle size. The UPC code 305361936723 can be found with the bar code at the bottom of the box. The affected lot numbers are: 09002 09379 10272 10368 10487 09131 09394 10273 10406 11058 09215 10154 10366 10433 The lot numbers can be found stamped into the bottom of the carton with the expiration date and above the label on the bottle printed in black. [Learn More]
About 898,000 units were affected by this recall.
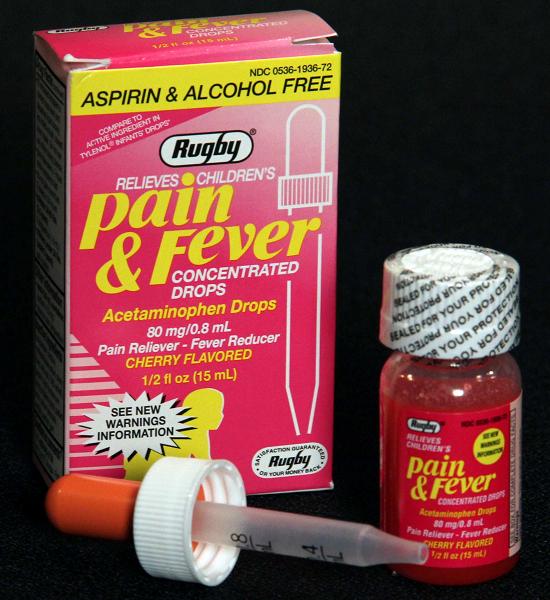
Rugby Children's Pain & Fever Concentrated Drops Recalled Due To Failure to Meet Child-Resistant Closure Requirement
.
Children’s Pain & Fever Concentrated Drops Recall Information
| Country of Origin | United States |
| Recall Date | 06/23/2011 |
| Recall ID | 705 |
| Recall Number | 11258 |
| Product Type | Liquid Drugs |
| Hazard | This over the counter medicine contains acetaminophen which calls for child-resistant packaging as required by the Poison Prevention Packaging Act. Although the original bottle has child-resistant packaging, a separate dropper unit provided for dispensing the drug to children does not. When in use, a child can access the medicine, posing serious health problems or death if more than the recommended dosage is consumed. |
| Injuries | None reported |
| Remedy | Consumers should immediately store this product with the child-resistant closure in place and keep it out of the reach of children. To arrange for a free replacement dropper, contact Altaire Pharmaceuticals at (800) 258-2471 9 a.m. to 5 p.m. ET Monday through Friday. |
| Remedy Option | Replace |
| Contact | For additional information, contact Rugby Laboratories at (800) 645-2158 between 9 a.m. and 5 p.m. ET Monday through Friday. |
| Last Updated | 07/08/2016 |
| Similar To |
